Titration is a common name in the chemistry field and acid-base titration lab is the most common subject among the titrations. Titration is a very common process in chemistry. This particular process is used to determine the concentration of an unknown solution. There are many kinds of titration among them acid-base titration is taught at the school level. So now, we are going to discuss a to z of the acid-base titration lab, acid-base titration theory, the acid-base titration lab answers, acid-base titration procedure, which means almost everything about the acid-base titration lab.
What does titration mean in Chemistry?
Titration is a very common word as well as a very common and popular method or technique in the chemistry lab. Now I can understand that you may know what is titration in terms of the chemistry lab, though I am trying to explain the definition of the word titration.
The titration is a laboratory process in which we can determine the concentration of a solution with the help of another solution of known concentration by reacting the two solutions in presence of indicator(s) in a proper procedure.
How does this technique work? – acid-base titration lab theory
To learn how this process work, we will use an example of an acid-base reaction. The most common acid and base (NaOH and HCl) are taken for the acid-base reaction. So the acid-base reaction is-
NaOH + HCl → NaCl + H2O
As the acid and base react to each other and neutralize each other, therefore the concentration and volume of the acid and base must be the same to be completely neutralized to form water and salt. So, we can see-
(volume × concentration) of acid = (volume × concentration) of base
Here, if we consider the-
- Volume of acid = Va
- Concentration of acid = Sa
- Volume of base = Vb
- Concentration of base = Sb
Then the equation will be- Va × Sa = Vb × Sb
But when we consider an acid and a base of different acidity and basicity respectively, then the problem will occur. Because the neutralization power of the acid and base are different. In this case, we will multiply the volume of acid by its basicity and the volume of the base by its acidity. I think u may get confused, so let’s make it simple- at first, we balance the chemical reaction of the acid-base neutralization reaction. Then, we divide the product of volume and concentration of acid by the number of moles of acid needed as well as volume and concentration of base by the number of moles of base needed. Let’s have a look at an example of such a reaction-
NaOH + H2SO4 → Na2SO4 + H2O
after balancing the acid-base reaction-
2NaOH + H2SO4 → Na2SO4 + 2H2O
The acid needed one mole and the base needed two moles. So the above equation for this reaction will be-
(Va × Sa)÷1 = (Vb × Sb)÷2
⇒ 2(Va × Sa) = Vb × Sb
I hope you become clear about how the acid-base titration technique work to determine the concentration of a solution with the help of a solution of known concentration.
Now I am going to tell you the details about how to work in a laboratory to perform a perfect titration to determine the concentration of a solution. But, first is, you have to know the chemistry laboratory instruments need to experiment.
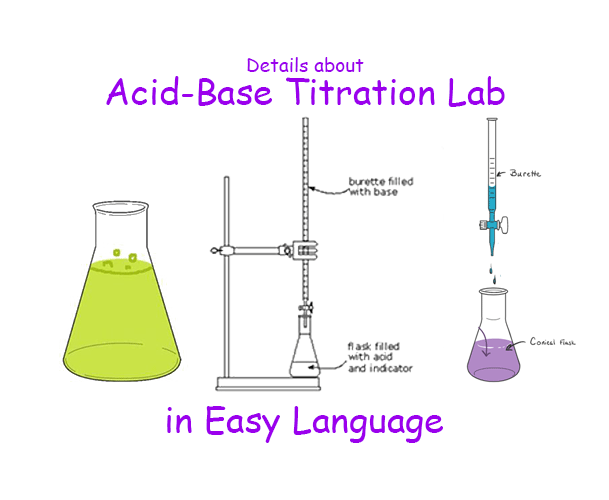
Instruments Need for Acid-Base Titration lab
As the lab is simple so we just need 3 instruments-
- A Burette (50mL is enough),
- A pipette (volume according to your lab process.),
- A conical flask
- Burette holding Stand, and
- A pipette filler.

Chemicals need for Acid-base titration lab
- Titrant
- Titrate
- Proper indicator(s)
The Titrant (acid or base) is the solution of which concentration is to be determined and Titrate (acid or base) is the solution of known concentration.
Acid-base titration lab process
- Make sure all the glass instruments are washed and dried properly.
- The volume of the titrant should be measured and taken with the help of a pipette into a conical flask.
- Then two to four drops of the indicator have to be added to the conical flask.
- The Burette has to fill with the titrate and the volume of the titrate has to note. This is the initial reading of the burette
- After that, the stopping crock of the burette has to loosen such that the titrate in the burette started to come out drop by drop per second and fall into the conical flask containing titrant.
- The conical flask should shake continuously as the burette running.
- Once all the titrant in the conical flask reacts with the titrate and produces neutral salt and water. Therefore, at this point in the conical flask, there is no acid as well as the base.
- At this stage, the indicator changes the color of the solution.
- At the same time, the stopping crock of the burette should be turned off immediately.
- Then the volume of the titrate in the burette has again to note. This is the final reading of the burette.
- The above process should repeat at least three times.

How to calculate the Acid-base titration lab data?
From the above discussion, we know that to determine the concentration of a solution by titration we need the volume of both acid and base and concentration of any one of them.
We get the measurement of volumes from the pipette and burette reading (Final burette reading – Initial Burette reading) and the concentration must be known as we have to prepare the other solution by our self. Now we have done our job. At this stage, we have to put the data into the equation as I mentioned above, and by doing a very simple calculation we get the concentration of other solution.
Precaution while working in a real Chemistry lab
- Always lab cote wearing
- Using high-quality gloves
- Using protective glass for eyes
- Wearing shoes (better to wear shoe cover).

Follow us on Twitter, Facebook, Linkedin, and Tumblr
You may like to read

Leave a Reply