Conformation and Reactivity in Acyclic compounds
The term conformation is used to denote any one of the infinite numbers of momentary arrangements of the atoms in space that results from rotation about single bonds.


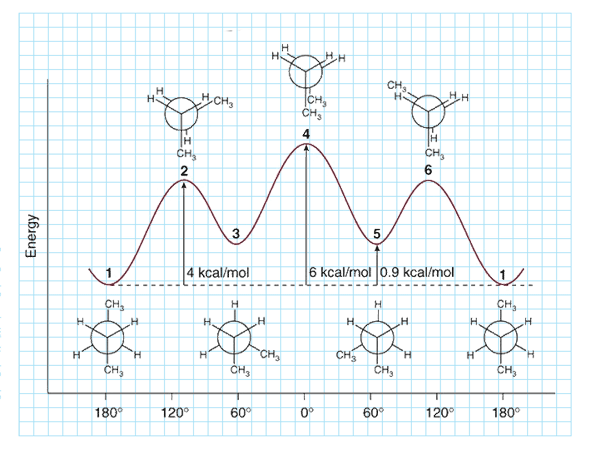
It is now recognized that the differences in energies between staggered and eclipsed are in some way associated with the interaction of the electron clouds in the C–H and C-C bonds.
Determination of configuration of cis-trans Isomers
Conformation & Physical Properties
Thermodynamic properties e.g. enthalpy, entropy, and related experimental properties e.g. specific heat, the heat of formation & several other physically measurable properties are dependent on conformation. In turn, their measurement may be used to determine conformation.
The physical methods are; IR, Raman, microwave, NMR spectra, dipole moment measurements, & data derived from electron diffraction & X-ray diffraction.
- Dipole moments: 1,2-dibromoethane

In the anti-C-Br bond is antiparallel & μ =0, whereas in the gauche form it is finite (µ>0). The actual µ is approx. 1 Debye (D) indicates that the molecule cannot exist entirely in the anti form. The fact that µ varies with temperature indicates that the molecule cannot exist entirely in gauche form. Two explanations; 1) Either anti & gauche forms are in equilibrium ( fig.6.5) & the equilibrium constants vary with temperature 2) or the molecule exists in either form but librates ( torsional oscillation ie. twisting to and fro about the bond in question, giving dipole moment ) around the C –C bond. The µ of the diastereoisomeric stilbene dichloride is incompatible with free rotation ( ie. there is a barrier).


- Once the principle here is recognized, the argument may be reversed and used to decide on which μ value belongs to which isomer ( diastereomer ).
- μ measurement rules out free rotation ( rotation is not free) but cannot give unequivocal proof of the existence of several conformers in equilibrium.
Definite evidence for the existence of several conformations comes from IR & Raman spectra ( at least for liquids & gas). Molecules e.g. 1,2-dibromoethane shows more than the allowed no of lines in the gas or liquid phase in its IR & Raman spectra. All the lines found in the solid-state spectra are also found in the gas or liquid state but additional lines appear in the latter state.
Strength from Weakness:
Conformational divergence of replaced cyclitols facilitated by CH … O hydrogen bonding between solid and solution states.
IR spectrum of ethylene glycol soln( dil) in CCl4 shows two bands.
- 3644 cm-1 ( due to unbonded OH)
- 3612cm-1 ( intramolecularly H-bonded OH)
Now H- bonded OH band is very strong means a considerable fraction of the molecules are in the gauche form despite the steric & dipolar repulsion of the OH group.

Follow Us On Facebook, Twitter, Linkdin, and Youtube.
Read More About:
Determination of configuration of cis-trans Isomers chemical method

Leave a Reply