Disclaimer:
This article is intended for educational and informational purposes only. It does not offer medical advice, promote, or sell pharmaceutical products. For any pharmaceutical manufacturing procedures, please consult certified industry professionals.
At first, I assure you that-“This content is intended for educational purposes only and does not promote or sell pharmaceutical products.”
Tablet manufacturing process
The tablet manufacturing process is a method of confectionery manufacture that shares many similarities with tablet pharmaceutical production. The manufacture of oral solid dosage forms such as tablets is a complex, multi-stage process in which the starting materials change their physical characteristics several times before the final dosage form is produced.
Tableting methods :
- Direct compression: suitable for medication with excellent flowability and compressibility that can be blended well and does not involve further granulation measures before the tablet press is introduced
- Dry granulation: suitable for moisture- and heat-sensitive medicines
- Wet granulation: Appropriate for humidity and heat-stable medicines.
−The first stage, milling & blending, is the same regardless of whether tablets are produced by direct compression or granulation; later steps vary.
−The production of tablets involves numerous unit procedures:
- Particle size reduction and Sizing,
- Blending,
- Granulation,
- Drying,
- Compression,
- (frequently) Coating
Tablet manufacturing process Flow chart

Dispensing (weighing and measuring)
−In any pharmaceutical manufacturing method, dispensing is the first phase.
−In pharmaceutical manufacturing, dispensing is also one of the most critical measures: during this phase, the weight of each component in the blend is determined by dose.
−Issues like:
- weighing accuracy,
- Manual handling of dust (laminar air stream booths, glove boxes),
- A lot of control over each ingredient,
- During dispensation, consideration should be given to material movement into and out of the dispensary.
Sizing
−The sizing (size reduction, milling, crushing, grinding, pulverization) is an important step (unit operation) involved in tablet manufacturing.
−Mixing or blending several solid pharmaceutical ingredients to manufacture compressed tablets is easier and more uniform when the ingredients are about the same size.
Advantages associated with size reduction in tablet manufacturing:
- It raises the region of the surface, which can increase the rate of active dissolution and thus bioavailability.
- We improved the uniformity of tablet-to-tablet content due to enhanced particle count per unit weight.
- Improved raw material flow properties.
- Enhanced dispersion of color and/or active ingredient in tablet excipients.
Drying
−Drying plays a crucial role in shaping the final quality and stability of pharmaceutical tablets.
| Concerns | Equipment | Purpose |
|
|
|
Stages of Tablet Formation (Compaction Cycle)
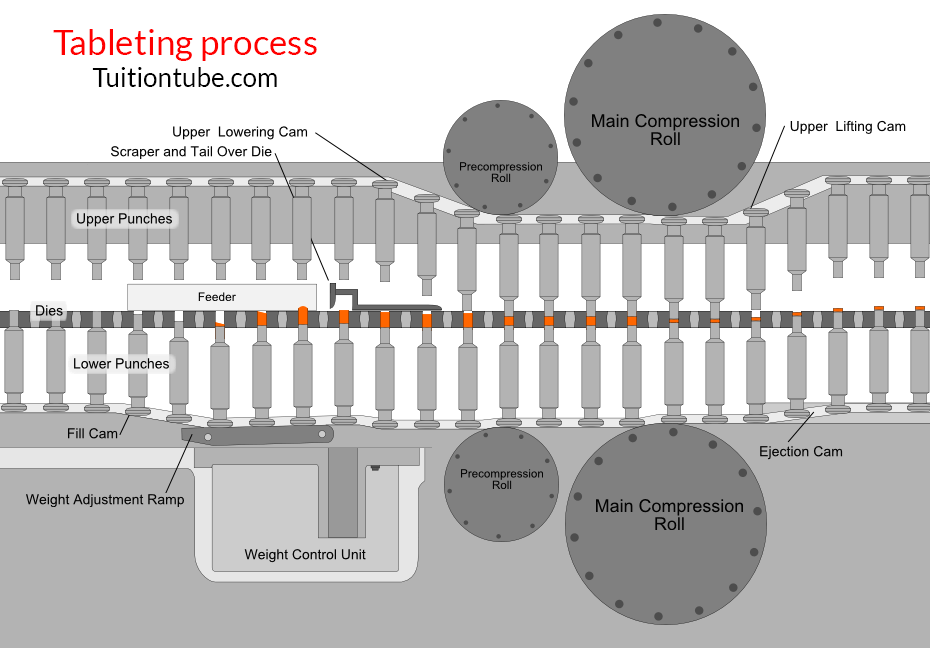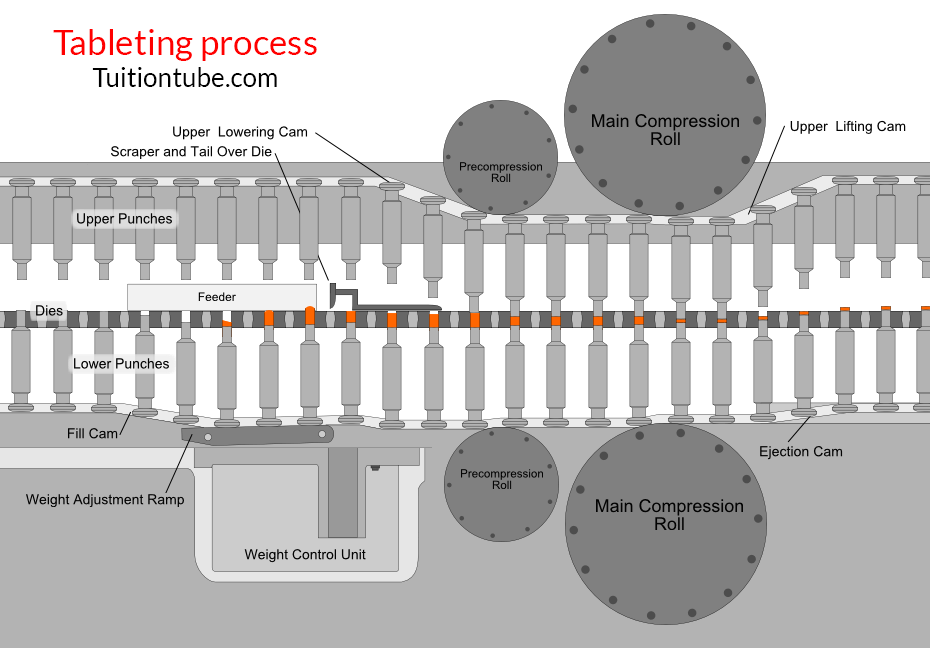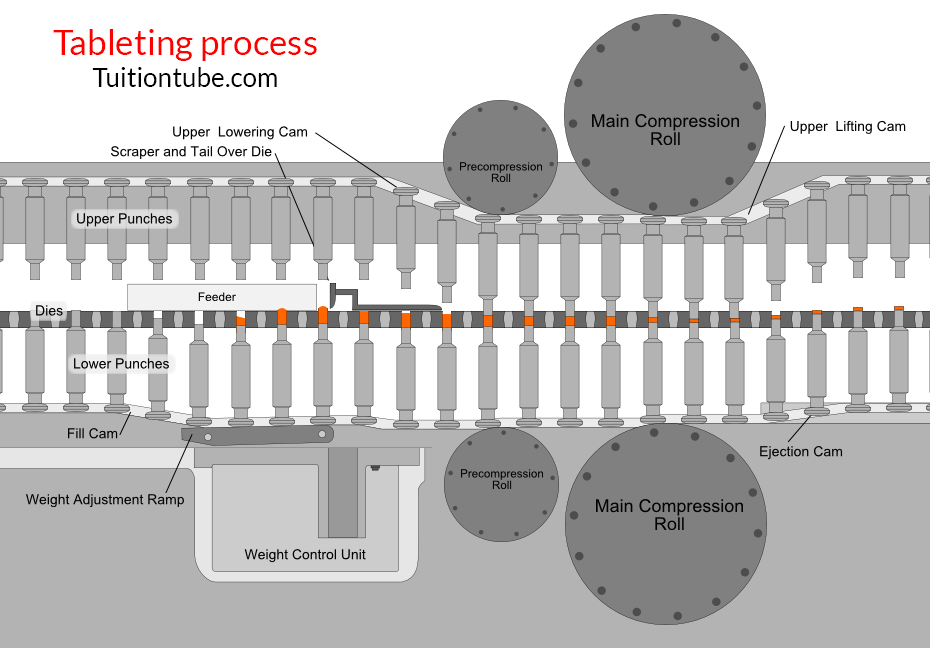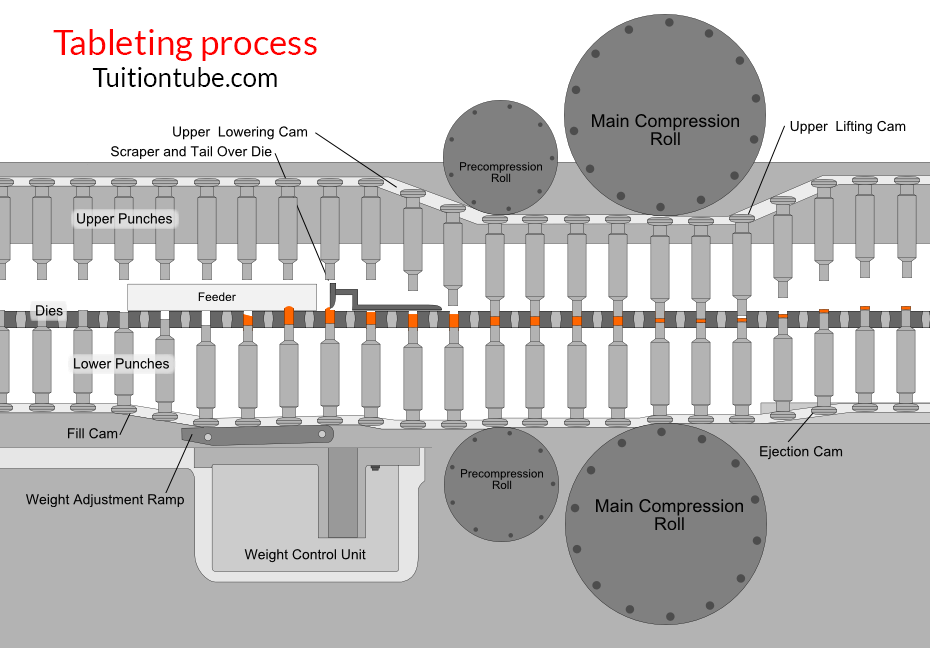
1- Die filling
The hopper’s gravitational flow of powder through the die table into the die. (The die is closed by the reduced punch at its bottom.)
2- Tablet formation
The upper punch comes down, reaches the die, and compresses the powder until a tablet is created.
-The upper punch leaves the powder, i.e., the compression stage, after reaching the highest applied force.
3- Tablet ejection
The smaller punch increases until the top of the die reaches its tip.
The tablet is then removed by a pushing machine from the die and the die table.
Follow Us On Facebook, Twitter, Linkdin, and YouTube.
Read More About:
The Michaelis-Menten Equation in biochemistry
Ionic Bond and Ionic Bond Formation, Definition, Properties in Chemistry


Leave a Reply